Fit adsorption data based on ion exchange reaction models.
Display solid & liquid & gas phase speciation diagrams as a funciton of pH & redox conditions.
| Alfisol, Item 007: | IExFit, version 3.4 Fit adsorption data based on ion exchange reaction models. Display solid & liquid & gas phase speciation diagrams as a funciton of pH & redox conditions. |
|
Software Overview |
|
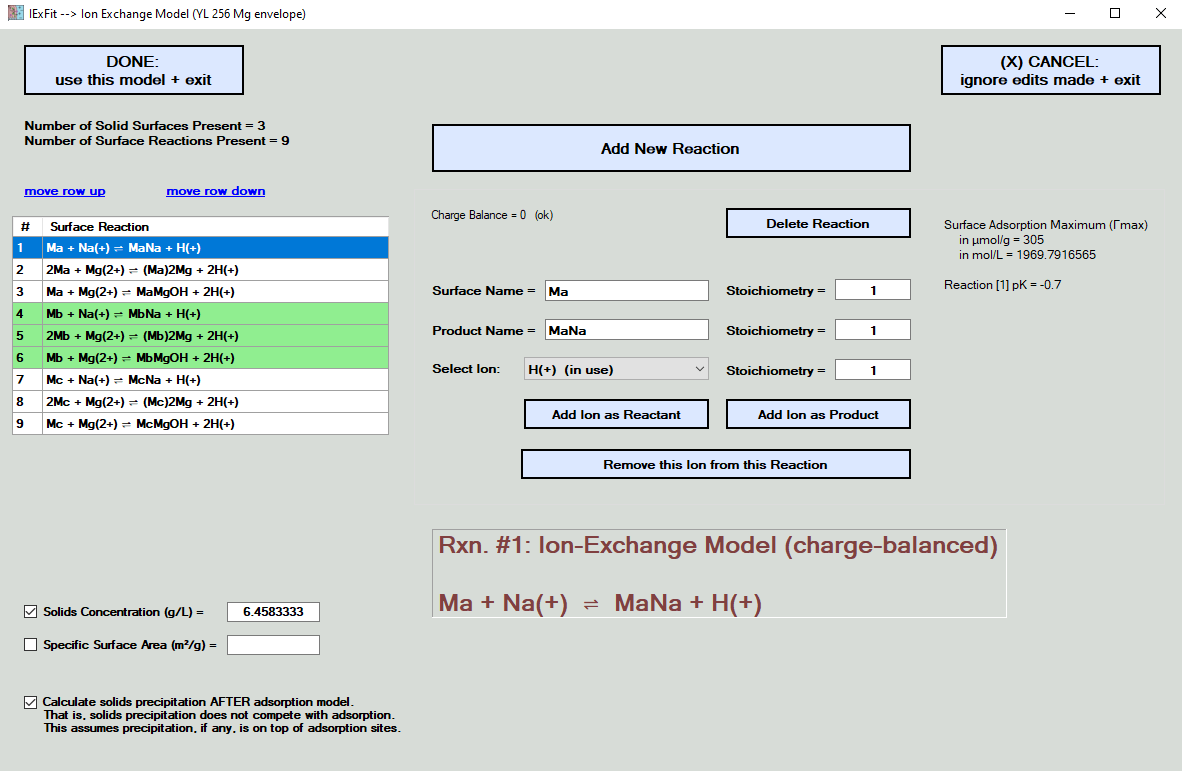
Here you enter the ion-exchange model that IExFit needs to optimize and fit your data.
In the example shown below, 9 surface reactions have been entered and three surfaces (which were labeled Ma, Mb, and Mc in this particular example).
Model results will be displayed in mol/L units, or similar units. If you offer information on solids concentration (optional), you can also display in mol/g units, or similar units. If you also offer information on specific surface area (optional), you can also display in μmol/m2 units.
You can add or edit a reaction surface site, as shown below. Note that the solids concentration and the specific surface area are not required, but they are needed if you later wish to display data or modeling results in mass or surface area units. Note that all calculations are made on mol/L units, which are then converted to the desired display units.
The example exercise below also allows precipitation reactions to occur. When this happens, an option appears that allows you to control if precipitation of solids occurs after the adsorption process is complete. If this option is not check, then precipitation occurs concurrently (and competitively) with the adsorption reactions. One reason to select this option is to assume that precipitation occurs on top of the adsorption sites. You cannot have desorption of species if precipitation is blocking it from the liquid phase. You also assume here that the adsorption reaction is faster than the precipitation reaction, which is a perfectly reasonable assumption considering that nucleation must occur prior to precipitation and surface seeding generally lowers the energy barriers involved. Thus, in the example shown below, there is no competition by precipitation reactions with adsorption reactions.