Fit adsorption data based on ion exchange reaction models.
Display solid & liquid & gas phase speciation diagrams as a funciton of pH & redox conditions.
| Alfisol, Item 007: | IExFit, version 3.4 Fit adsorption data based on ion exchange reaction models. Display solid & liquid & gas phase speciation diagrams as a funciton of pH & redox conditions. |
|
Software Overview |
|
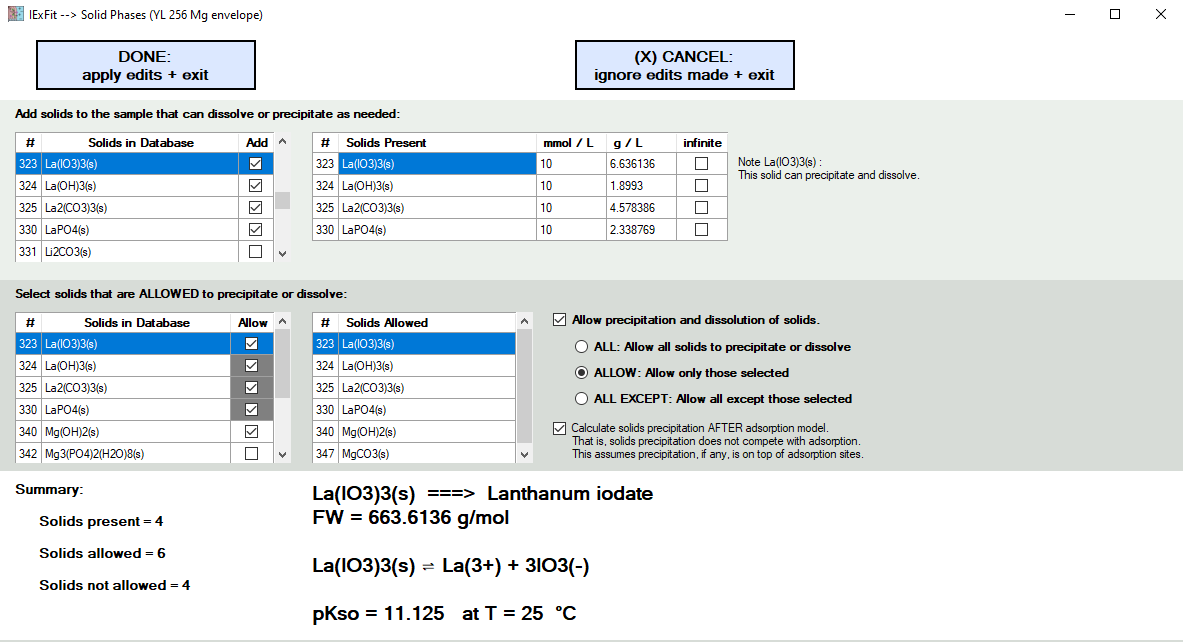
In the lower section here, you decide if solids are allowed to precipitate. If so, you also decide which ones are allowed or not allowed.
In the upper section here, you have the option to specify that solids exist in the experiment as initial conditions. If so, you also state how much is present either in mmol/L units or g/L units. You can also select "infinite" for hypothetical exercises. The formula weight (FW) and the reaction involved are shown in the bottom panel to help you select the correct solid species.
In the example shown below, six solids were selected as allowed to exist. Of these six, four of them must be allowed because they were also selected as solids initially present in the top panel. IExFit will not allow you to uncheck them as long as they are also selected in the top panel as "solids present".
IExFit will dissolve or precipitate these solid species as needed for each pH condition in the exercise.
The example below also involves an adsorption model. When this happens, an option appears that allows you to control if precipitation of solids occurs after the adsorption process is complete. If this option is not check, then precipitation occurs concurrently (and competitively) with the adsorption reactions. One reason to select this option is to assume that precipitation occurs on top of the adsorption sites. You cannot have desorption of species if precipitation is blocking it from the liquid phase. You also assume here that the adsorption reaction is faster than the precipitation reaction, which is a perfectly reasonable assumption considering that nucleation must occur prior to precipitation and surface seeding generally lowers the energy barriers involved. Thus, in the example shown below, there is no competition by precipitation reactions with adsorption reactions.